Abstract
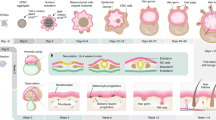
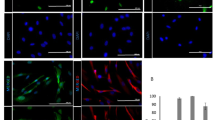
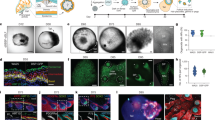
This protocol describes methods of isolating skin-derived precursors (SKPs) from rodent and human skin, and for generating and enriching Schwann cells from rodent SKPs. SKPs are isolated as a population of non-adherent cells from the dermis that proliferate and self-renew as floating spheres in response to fibroblast growth factor 2 (FGF2) and epidermal growth factor (EGF). Their differentiation into Schwann cells and subsequent enrichment of these differentiated progeny involves culturing SKPs as adherent cells in the absence of FGF2 and EGF, but in the presence of neuregulins, and then mechanically isolating the Schwann cell colonies using cloning cylinders. Methods for expanding and characterizing these Schwann cells are provided. Generation of primary SKPs takes approximately 2 weeks, while differentiation of Schwann cells requires an additional 4–6 weeks.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Aguayo, A.J., Kasarjian, J., Skamene, E., Kongshavn, P. & Bray, G.M. Myelination of mouse axons by Schwann cells transplanted from normal and abnormal human nerves. Nature 268, 753–755 (1977).
David, S. & Aguayo, A.J. Axonal regeneration after crush injury of rat central nervous system fibres innervating peripheral nerve grafts. J. Neurocytol. 14, 1–12 (1985).
Pearse, D.D. et al. Transplantation of Schwann cells and olfactory ensheathing glia after spinal cord injury: does pretreatment with methylprednisolone and interleukin-10 enhance recovery? J. Neurotrauma. 21, 1223–1239 (2004).
Pearse, D.D. et al. cAMP and Schwann cells promote axonal growth and functional recovery after spinal cord injury. Nat. Med. 10, 610–616 (2004).
Xu, X.M., Chen, A., Guenard, V., Kleitman, N. & Bunge, M.B. Bridging Schwann cell transplants promote axonal regeneration from both the rostral and caudal stumps of transected adult rat spinal cord. J. Neurocytol. 26, 1–16 (1997).
Xu, X.M., Zhang, S.X., Li, H., Aebischer, P. & Bunge, M.B. Regrowth of axons into the distal spinal cord through a Schwann-cell-seeded mini-channel implanted into hemisected adult rat spinal cord. Eur. J. Neurosci. 11, 1723–1740 (1999).
Pinzon, A., Calancie, B., Oudega, M. & Noga, B.R. Conduction of impulses by axons regenerated in a Schwann cell graft in the transected adult rat thoracic spinal cord. J. Neurosci. Res. 64, 533–541 (2001).
Paino, C.L. & Bunge, M.B. Induction of axon growth into Schwann cell implants grafted into lesioned adult rat spinal cord. Exp. Neurol. 114, 254–257 (1991).
Kocsis, J.D., Akiyama, Y., Lankford, K.L. & Radtke, C. Cell transplantation of peripheral-myelin-forming cells to repair the injured spinal cord. J. Rehabil. Res. Dev. 39, 287–298 (2002).
Keirstead, H.S., Morgan, S.V., Wilby, M.J. & Fawcett, J.W. Enhanced axonal regeneration following combined demyelination plus Schwann cell transplantation therapy in the injured adult spinal cord. Exp. Neurol. 159, 225–236 (1999).
Fouad, K. et al. Combining Schwann cell bridges and olfactory-ensheathing glia grafts with chondroitinase promotes locomotor recovery after complete transection of the spinal cord. J. Neurosci. 25, 1169–1178 (2005).
Barakat, D.J. et al. Survival, integration, and axon growth support of glia transplanted into the chronically contused spinal cord. Cell Transplant. 14, 225–240 (2005).
Li, R. Culture methods for selective growth of normal rat and human Schwann cells. Methods Cell Biol. 57, 167–186 (1998).
Mason, P.W., Attema, B.L. & DeVries, G.H. Isolation and characterization of neonatal Schwann cells from cryopreserved rat sciatic nerves. J. Neurosci. Res. 31, 731–744 (1992).
Oda, Y., Okada, Y., Katsuda, S., Ikeda, K. & Nakanishi, I. A simple method for the Schwann cell preparation from newborn rat sciatic nerves. J. Neurosci. Methods 28, 163–169 (1989).
Peulve, P., Laquerriere, A., Paresy, M., Hemet, J. & Tadie, M. Establishment of adult rat Schwann cell cultures: effect of β-FGF, α-MSH, NGF, PDGF, and TGF-β on cell cycle. Exp. Cell Res. 214, 543–550 (1994).
Vroemen, M. & Weidner, N. Purification of Schwann cells by selection of p75 low affinity nerve growth factor receptor expressing cells from adult peripheral nerve. J. Neurosci. Methods 124, 135–143 (2003).
Assouline, J.G., Bosch, E.P. & Lim, R. Purification of rat Schwann cells from cultures of peripheral nerve: an immunoselective method using surfaces coated with anti-immunoglobulin antibodies. Brain Res. 277, 389–392 (1983).
Casella, G.T., Bunge, R.P. & Wood, P.M. Improved method for harvesting human Schwann cells from mature peripheral nerve and expansion in vitro. Glia 17, 327–338 (1996).
Kreider, B.Q. et al. Enrichment of Schwann cell cultures from neonatal rat sciatic nerve by differential adhesion. Brain Res. 207, 433–444 (1981).
Moretto, G., Kim, S.U., Shin, D.H., Pleasure, D.E. & Rizzuro, N. Long-term cultures of human adult Schwann cells isolated from autopsy materials. Acta Neuropathol. (Berl) 64, 15–21 (1984).
Scarpini, E., Kreider, B.Q., Lisak, R.P. & Pleasure, D.E. Establishment of Schwann cell cultures from adult rat peripheral nerves. Exp. Neurol. 102, 167–176 (1988).
Morrissey, T.K., Kleitman, N. & Bunge, R.P. Isolation and functional characterization of Schwann cells derived from adult peripheral nerve. J. Neurosci. 11, 2433–2442 (1991).
Rutkowski, J.L., Tennekoon, G.I. & McGillicuddy, J.E. Selective culture of mitotically active human Schwann cells from adult sural nerves. Ann. Neurol. 31, 580–586 (1992).
Rutkowski, J.L., Kirk, C.J., Lerner, M.A. & Tennekoon, G.I. Purification and expansion of human Schwann cells in vitro. Nat. Med. 1, 80–83 (1995).
Toma, J.G. et al. Isolation of multipotent adult stem cells from the dermis of mammalian skin. Nat. Cell Biol. 3, 778–784 (2001).
Fernandes, K.J.L. et al. A dermal niche for multipotent adult skin-derived precursor cells. Nat. Cell Biol. 6, 1082–1093 (2004).
Fernandes, K.J. et al. Analysis of the neurogenic potential of multipotent skin-derived precursors. Exp. Neurol. 201, 32–48 (2006).
Toma, J.G., McKenzie, I.A., Bagli, D. & Miller, F.D. Isolation and characterization of multipotent skin-derived precursors from human skin. Stem Cells 23, 727–737 (2005).
Joannides, A. et al. Efficient generation of neural precursors from adult human skin: astrocytes promote neurogenesis from skin-derived stem cells. Lancet 364, 172–178 (2004).
McKenzie, I.A., Biernaskie, J., Toma, J.G., Midha, R. & Miller, F.D. Skin-derived precursors generate myelinating Schwann cells for the injured and dysmyelinated nervous system. J. Neurosci. 26, 6651–6660 (2006).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr. Freda Miller is a consultant for Aggregate Therapeutics, a biotechnology company who has licensed patents covering SKPs technology from McGill University, who owns the technology.
Rights and permissions
About this article
Cite this article
Biernaskie, J., McKenzie, I., Toma, J. et al. Isolation of skin-derived precursors (SKPs) and differentiation and enrichment of their Schwann cell progeny. Nat Protoc 1, 2803–2812 (2006). https://doi.org/10.1038/nprot.2006.422
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2006.422
This article is cited by
-
Implantation of skin-derived precursor Schwann cells improves erectile function in a bilateral cavernous nerve injury rat model
Basic and Clinical Andrology (2023)
-
Expandable Lung Epithelium Differentiated from Human Embryonic Stem Cells
Tissue Engineering and Regenerative Medicine (2022)
-
Improvement of sensory neuron growth and survival via negatively regulating PTEN by miR-21-5p-contained small extracellular vesicles from skin precursor-derived Schwann cells
Stem Cell Research & Therapy (2021)
-
Plant callus-derived shikimic acid regenerates human skin through converting human dermal fibroblasts into multipotent skin-derived precursor cells
Stem Cell Research & Therapy (2021)
-
Advances and clinical challenges for translating nerve conduit technology from bench to bed side for peripheral nerve repair
Cell and Tissue Research (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.