Abstract
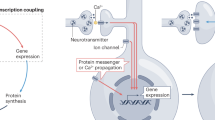
Information storage in the nervous system requires transcription triggered by synaptically evoked calcium signals. It has been suggested that translocation of calmodulin into the nucleus, initiated by submembranous calcium transients, relays synaptic signals to CREB. Here we show that in hippocampal neurons, signaling to CREB can be activated by nuclear calcium alone and does not require import of cytoplasmic proteins into the nucleus. The nucleus is particularly suited to integrate neuronal firing patterns, and specifies the transcriptional outputs through a burst frequency-to-nuclear calcium amplitude conversion. Calcium release from intracellular stores promotes calcium wave propagation into the nucleus, which is critical for CREB-mediated transcription by synaptic NMDA receptors. Pharmacological or genetic modulation of nuclear calcium may directly affect transcription-dependent memory and cognitive functions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Milner, B., Squire, L. R. & Kandel, E. R. Cognitive neuroscience and the study of memory. Neuron 20, 445–468 (1998).
Ghosh, A. & Greenberg, M. E. Calcium signaling in neurons: molecular mechanisms and cellular consequences. Science 268, 239–247 (1995).
Hardingham, G. E. & Bading, H. Calcium as a versatile second messenger in the control of gene expression. Microsc. Res. Tech. 46, 348–355 (1999).
Bailey, C. H., Bartsch, D. & Kandel, E. R. Toward a molecular definition of long-term memory storage. Proc. Natl. Acad. Sci. USA 93, 13445–13452 (1996).
Silva, A. J., Kogan, J. H., Frankland, P. W. & Kida, S. CREB and memory. Annu. Rev. Neurosci. 21, 127–148 (1998).
Ahn, S., Riccio, A. & Ginty, D. D. Spatial considerations for stimulus-dependent transcription in neurons. Annu. Rev. Physiol. 62, 803–823 (2000).
Adams, J. P., Roberson, E. D., English, J. D., Selcher, J. C. & Sweatt, J. D. MAPK regulation of gene expression in the central nervous system. Acta. Neurobiol. Exp. (Warsz.) 60, 377–394 (2000).
Bading, H. Transcription-dependent neuronal plasticity: The nuclear calcium hypothesis. Eur. J. Biochem. 267, 5280–5283 (2000).
Bading, H. & Greenberg, M. E. Stimulation of protein tyrosine phosphorylation by NMDA receptor activation. Science 253, 912–914 (1991).
Ginty, D. D., Bonni, A. & Greenberg, M. E. Nerve growth factor activates a Ras-dependent protein kinase that stimulates c-fos transcription via phosphorylation of CREB. Cell 77, 713–725 (1994).
Xing, J., Ginty, D. D. & Greenberg, M. E. Coupling of the RAS-MAPK pathway to gene activation by RSK2, a growth factor-regulated CREB kinase. Science 273, 959–963 (1996).
Impey, S. et al. Cross talk between ERK and PKA is required for Ca2+ stimulation of CREB-dependent transcription and ERK nuclear translocation. Neuron 21, 869–883 (1998).
Chawla, S., Hardingham, G. E., Quinn, D. R. & Bading, H. CBP: a signal-regulated transcriptional coactivator controlled by nuclear calcium and CaM kinase IV. Science 281, 1505–1509 (1998).
Sheng, M., Thompson, M. A. & Greenberg, M. E. CREB: a Ca2+-regulated transcription factor phosphorylated by calmodulin-dependent kinases. Science 252, 1427–1430 (1991).
Cruzalegui, F. H. & Means, A. R. Biochemical characterization of the multifunctional calcium/calmodulin dependent protein kinase type IV expressed in insect cells. J. Biol. Chem. 268, 26171–26178 (1993).
Sun, P., Enslen, H., Myung, P. S. & Maurer, R. A. Differential activation of CREB by Ca2+/calmodulin-dependent protein kinases type II and type IV involves phosphorylation of a site that negatively regulates activity. Genes Dev. 8, 2527–2539 (1994).
Matthews, R. P. et al. Calcium/calmodulin-dependent protein kinase types II and IV differentially regulate CREB-dependent gene expression. Mol. Cell. Biol. 14, 6107–6116 (1994).
Bito, H., Deisseroth, K. & Tsien, R. W. CREB phosphorylation and dephosphorylation: a Ca2+ and stimulus duration-dependent switch for hippocampal gene expression. Cell 87, 1203–1214 (1996).
Hardingham, G. E., Chawla, S., Cruzalegui, F. H. & Bading, H. Control of recruitment and transcription-activating function of CBP determines gene regulation by NMDA receptors and L-type calcium channels. Neuron 22, 789–798 (1999).
Sheng, M., Dougan, S. T., McFadden, G. & Greenberg, M. E. Calcium and growth factor pathways of c-fos transcriptional activation require distinct upstream regulatory sequences. Mol. Cell. Biol. 8, 2787–2796 (1988).
Bonni, A., Ginty, D. D., Dudek, H. & Greenberg, M. E. Serine 133-phosphorylated CREB induces transcription via a cooperative mechanism that may confer specificity to neurotrophin signals. Mol. Cell. Neurosci. 6, 168–183 (1995).
Hu, S.-C., Chrivia, J. & Ghosh, A. Regulation of CBP-mediated transcription by neuronal calcium signaling. Neuron 22, 799–808 (1999).
Goodman, R. H. & Smolik, S. CBP/p300 in cell growth, transformation, and development. Genes Dev. 14, 1553–1577 (2000).
Cruzalegui, F. H., Hardingham, G. E. & Bading, H. c-Jun functions as a calcium-regulated transcriptional activator in the absence of JNK/SAPK1 activation. EMBO J. 18, 1335–1344 (1999).
Deisseroth, K., Heist, E. K. & Tsien, R. W. Calmodulin translocation to the nucleus supports CREB phosphorylation in hippocampal neurons. Nature 392, 198–202 (1998).
Pruschy, M., Ju, Y., Spitz, L., Carafoli, E. & Goldfarb, D. S. Facilitated nuclear transport of calmodulin in tissue culture cells. J. Cell Biol. 127, 1527–1536 (1994).
Lerea, L. S., Butler, L. S. & McNamara, J. O. NMDA and non–NMDA receptor-mediated increase of c-fos mRNA in dentate gyrus neurons involved calcium influx via different routes. J. Neurosci. 12, 2973–2981 (1992).
Bading, H., Ginty, D. D. & Greenberg, M. E. Regulation of gene expression in hippocampal neurons by distinct calcium signaling pathways. Science 260, 181–186 (1993).
Hardingham, G. E., Chawla, S., Johnson, C. M. & Bading, H. Distinct functions of nuclear and cytoplasmic calcium in the control of gene expression. Nature 385, 260–265 (1997).
Fields, R. D., Esthete, F., Stevens, B., & Itoh, K. Action potential-dependent regulation of gene expression: temporal specificity in calcium, cAMP-reponsive element binding proteins, and mitogen-activated protein kinase signalling. J. Neurosci. 17, 7252–7266 (1997).
Ginty, D. D. et al. Regulation of CREB phosphorylation in the suprachiasmatic nucleus by light and a circadian clock. Science 268, 238–241 (1993).
Hagiwara, M. et al. Transcriptional attenuation following cAMP induction requires PP-1-mediated dephosphorylation of CREB. Cell 70, 105–113 (1992).
Lipp, P., Thomas, D., Berridge, M. J. & Bootman, M. D. Nuclear calcium signalling by individual cytoplasmic calcium puffs. EMBO J. 16, 7166–7173 (1997).
Nakazawa, H. & Murphy, T. H. Activation of nuclear calcium dynamics by synaptic stimulation in cultured cortical neurons. J. Neurochem. 73, 1075–1083 (1999).
Olwin, B. B., Edelman, A. M., Krebs, E. G. & Storm, D. R. Quantitation of energy coupling between Ca2+, calmodulin, skeletal muscle myosin light chain kinase, and kinase substrates. J. Biol. Chem. 259, 10949–10955 (1984).
Shaw, P., Schröter, H. & Nordheim, A. The ability of a ternary complex to form over the serum response element correlates with serum inducibility of the c-fos promoter. Cell 56, 563–572 (1989).
Treisman, R. Ternary complex factors: growth factor regulated transcriptional activators. Curr. Opin. Genet. Dev. 4, 96–101 (1994).
Johnson, C. M., Hill, C. S., Chawla, S., Treisman, R. & Bading, H. Calcium controls gene expression via three distinct pathways that can function independently of the Ras/Mitogen-activated protein kinases (ERKs) signaling cascade. J. Neurosci. 17, 6189–6202 (1997).
Magee, J. C. & Johnston, D. A synaptically controlled, associative signal for Hebbian plasticity in hippocampal neurons. Science 275, 209–213 (1997).
Markram, H., Lübke, J., Frotscher, M. & Sakmann, B. Regulation of synaptic efficacy by coincidence of postsynaptic APs and EPSPs. Science 275, 213–215 (1997).
Larkum, M. E., Zhu, J. J. & Sakmann, B. A new cellular mechanism for coupling inputs arriving at different cortical layers. Nature 398, 338–341 (1999).
Nakamura, T., Barbara, J. G., Nakamura, K. & Ross, W. N. Synergistic release of Ca2+ from IP3-sensitive stores evoked by synaptic activation of mGluRs paired with backpropagating action potentials. Neuron 24, 727–737 (1999).
Christie, B. R., Eliot, L. S., Ito, K., Miyakawa, H. & Johnston, D. Different Ca2+ channels in soma and dendrites of hippocampal pyramidal neurons mediate spike-induced Ca2+ influx. J. Neurophysiol. 73, 2553–2557 (1995).
Schiller, J., Helmchen, F. & Sakmann B. Spatial profile of dendritic calcium transients evoked by action potentials in rat neocortical pyramidal neurones. J. Physiol. (Lond.) 487, 583–600 (1995).
Frey, U. & Morris, R. G. Synaptic tagging and long-term potentiation. Nature 385, 533–536 (1997).
Dolmetsch, R. E., Lewis, R. S., Goodnow, C. C. & Healy, J. I. Differential activation of transcription factors induced by calcium response amplitude and duration. Nature 386, 855–858 (1997).
Deyo, R. A., Straube, K. T. & Disterhoft, J. F. Nimodipine facilitates associative learning in aging rabbits. Science 243, 809–811 (1989).
Levere, T. E. & Walker, A. Old age and cognition: enhancement of recent memory in aged rats by the calcium channel blocker nimodipine. Neurobiol. Aging 13, 63–66 (1991).
Sandin, M., Jasmin, S. & Levere, T. E. Aging and cognition: facilitation of recent memory in aged nonhuman primates by nimodipine. Neurobiol. Aging 11, 573–575 (1990).
Morich, F. J. et al. Nimodipine in the treatment of probable Alzheimer's disease—results of two multicentre trials. Clin. Drug Invest. 11, 185–195 (1996).
Acknowledgements
We thank N.R. Hardingham and J. Jack for discussion and background electrophysiological data, E. Gruenstein for advice on the 4-AP experiments, and B. Wisden for discussion. This work was supported by the MRC, Dunhill Medical Trust, The Royal College of Surgeons of England, the Sackler Medical Research Centre and Clare College, Cambridge.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hardingham, G., Arnold, F. & Bading, H. Nuclear calcium signaling controls CREB-mediated gene expression triggered by synaptic activity. Nat Neurosci 4, 261–267 (2001). https://doi.org/10.1038/85109
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/85109
This article is cited by
-
Deletion of voltage-gated calcium channels in astrocytes decreases neuroinflammation and demyelination in a murine model of multiple sclerosis
Journal of Neuroinflammation (2023)
-
Extrasynaptic NMDA receptors in acute and chronic excitotoxicity: implications for preventive treatments of ischemic stroke and late-onset Alzheimer’s disease
Molecular Neurodegeneration (2023)
-
Excitation–transcription coupling, neuronal gene expression and synaptic plasticity
Nature Reviews Neuroscience (2023)
-
Activation of CREB-BDNF Pathway in Pyramidal Neurons in the Hippocampus Improves the Neurological Outcome of Mice with Ischemic Stroke
Molecular Neurobiology (2023)
-
Integration of nuclear Ca2+ transients and subnuclear protein shuttling provides a novel mechanism for the regulation of CREB-dependent gene expression
Cellular and Molecular Life Sciences (2023)