Abstract
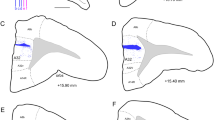
The different cytoarchitectonic regions of the medial prefrontal cortex (mPFC) have recently been shown to play divergent roles in associative learning in rabbits. To determine if these subareas of the mPFC, including areas 24 (anterior cingulate cortex), 25 (infralimbic cortex), and 32 (prelimbic cortex) have differential efferent connections with other cortical and subcortical areas in the rabbit, anterograde and retrograde tracing experiments were performed using thePhaseolus vulgaris leukoagglutinin (PHA-L), and horseradish peroxidase (HRP) techniques. All three areas showed local dorsal-ventral projections into each of the other areas, and a contralateral projection to the homologous area on the other side of the brain. All three also revealed a trajectory through the striatum, resulting in heavy innervation of the caudate nucleus, the claustrum, and a lighter projection to the agranular insular cortex. The thalamic projections of areas 24 and 32 were similar, but not identical, with projections to the mediodorsal nucleus (MD) and all of the midline nuclei. However, the primary thalamic projections from area 25 were to the intralaminar and midline nuclei. All three areas also projected to the ventromedial and to a lesser extent to the ventral posterior thalamic nuclei. Projections were also observed in the lateral hypothalamus, in an area just lateral to the descending limb of the fornix. Amygdala projections from areas 32 and 24 were primarily to the lateral, basolateral and basomedial nuclei, but area 25 also projected to the central nucleus. All three areas also showed projections to the midbrain periaqueductal central gray, median raphe nucleus, ventral tegmental area, substantia nigra, locus coeruleus and pontine nuclei. However, only areas 24 and the more dorsal portions of area 32 projected to the superior colliculus. Area 25 and the ventral portions of area 32 also showed a bilateral projection to the parabrachial nuclei and dorsal and ventral medulla. The dorsal portions of area 32, and all of area 24 were, however, devoid of these projections. It is suggested that these differential projections are responsible for the diverse roles that the cytoarchitectonic subfields of the mPFC have been demonstrated to play in associative learning.
Similar content being viewed by others
References
Arikuni T, Ban T Jr (1978) Subcortical afférents to the prefrontal cortex in rabbits Exp Brain Res 32:69–75
Bacon, SJ, Smith AD (1993) A monosynaptic pathway from an identified vasomotor centre in the medial prefrontal cortex to an autonomic area in the thoracic spinal cord. Neuroscience 54:719–728
Barbas H, Mesulam M (1985) Cortical afferent input to the principalis region of the Rhesus monkey. Neuroscience 15:619–637
Benjamin RM, Jackson JC, Golden GT (1978) Cortical projections of the thalamic mediodorsal nucleus in the rabbit. Brain Res 141:251–265
Berendse HW, Graaf YGD, Groenewegen HJ (1992) Topographical organization and relationship with ventral striatal compartments of prefrontal corticostriatal projections in the rat. J Comp Neurol 316:314–347
Brodmann K (1909) Vergleichende Lokalisationslehre der Grosshirnrinde in ihren Prinzipien dargestellt auf grund des Zellenbaues. Barth, Leipzig, Austria
Buchanan SL, Powell DA (1982a) Cingulate cortex: its role in Pavlovian conditioning. J Comp Physiol Psychol 96:755–774
Buchanan SL, Powell DA (1982b) Cingulate damage attentuates conditioned bradycardia. Neurosci Lett 29:261–268
Buchanan SL, Powell DA (1993) Cingulothalamic and prefrontal control of autonomic function. In: Vogt BA, Gabriel M (eds) Neurobiology of cingulate cortex and limbic thalamus. Birkhauser, Cambridge, MA, pp 381–414
Buchanan SL, Powell DA, Thompson RH (1989) Prefrontal projections to the medial nuclei of the dorsal thalamus in the rabbit. Neurosci Lett 106:55–59
Burns SM, Wyss JM (1985) The involvement of the anterior cingulate cortex in blood pressure control. Brain Res 340:71–77
Cassell MD, Wright DJ (1986) Topography of projections from the medial prefrontal cortex to the amygdala in the rat. Brain Res Bull 17:321–333
Damasio AR, Tranel D, Damasio H (1990) Individuals with sociopathic behavior caused by frontal damage fail to respond autonomically to social stimuli. Behav Brain Res 41:81–94
Fuster JM (1989) The prefrontal cortex: anatomy, physiology and neuropsychology of the frontal lobe (2nd edn). Raven Press, New York
Frysztak RJ, Neafsey EJ (1991) The effect of medial frontal cortex lesions on respiration, ‘freezing’, and ultrasonic vocalizations during conditioned emotional responses in rats. Cereb Cortex 1:418–425
Gentile CG, Jarrell TW, Teich A, McCabe PM, Schneiderman N (1986) The role of amygdaloid central nucleus in the retention of differential Pavlovian conditioning of bradycardia in rabbits. Behav Brain Res 20:263–273
Gibbs CM, Powell DA (1991) Single-unit activity in the dorsomedial prefrontal cortex during the expression of discriminative bradycardia in rabbits. Behav Brain Res 43:79–92
Goldman-Rakic PS (1990) Cellular and circuit basis of working memory in prefrontal cortex of nonhuman primates. Prog. Brain Res. 85:325–326
Goldman-Rakic PS, Porrino LJ (1985) The primate mediodorsal (MD) nucleus and its projection to the frontal lobe. J Comp Neurol 242:535–560
Groenewegen HJ, Berendse HW, Wolters JG, Lohman AHM (1990). The anatomical relationship of the prefrontal cortex with the striatopallidal system, the thalamus and the amygdala: evidence for a parallel organization. Prog Brain Res:95-118
Hurley KM, Herbert H, Moga MM, Saper CB (1991) Efferent projections of the infralimbic cortex of the rat. J Comp Neurol 308:249–276
Kaada BR (1960) Cingulate, posterior orbital, anterior insular, and temporal pole cortex. In: Field J, Magoun HW, Hall VE (eds) Handbook of physiology, sect 1 ; Neurophysiology vol 2 American Physiological Society, Washington DC, pp 1345–1372
Kapp BS, Frysinger RC, Gallagher M, Haselton JR (1979) Amygdala central nucleus lesions: effect on heart rate conditioning in the rabbit. Physiol Behav 23:1109–1117
Kapp BS, Schwaber JS, Driscoll PA (1985a) Frontal cortex projections to the amygdaloid central nucleus in the rabbit. Neuroscience 15:327–346
Kapp BS, Schwaber JS, Driscoll PA (1985b) The organization of insular cortex projections to the amygdaloid central nucleus and autonomic regulatory nuclei of the dorsal medulla. Brain Res 360:355–360
Kazis E, Milligan WL, Powell DA (1973) Autonomic-somatic relationships: blockade of heart rate and corneoretinal potential. J Comp Physiol Psychol 84:98–110
Kolb B (1984) Functions of the frontal cortex of the rat: a comparative review. Brain Res Rev 8:65–98
Kosmal A (1986) Topographical organization of frontal association cortex afférents originating in ventral thalamic nuclei in dog brain. Acta Neurobiol Exp 46:105–117
Krettek JE, Price JH (1977) The cortical projections of the mediodorsal nucleus and adjacent thalamic nuclei in the rat. J Comp Neurol 171:157–192
LeDoux JE, Iwata J, Cicchetti P, Reis DJ (1988) Different projections of the central amygdaloid nucleus mediate autonomie and behavioral correlates of conditioned fear. J Neurosci 8:2517–2529
LeDoux JE, Färb C, Ruggiero DA (1990) Topographic organization of neurons in the acoustic thalamus that project to the amygdala. J Neurosci 10:1043–1054
Leichnetz GR, Povlishock JT, Astruc J (1976) A prefronto-amygdaloid projection in the monkey: light and electron microscopic evidence. Neurosci Lett 2:261–266
Leonard CM (1969) The prefrontal cortex of the rat. I. Cortical projection of the mediodorsal nucleus II. Efferent connections. Brain Res 12:321–343
Markowitsch HJ, Pritzel M (1978) The insular region: part of the prefrontal cortex? Neurosci Biobehav Rev 2:271–276
Markowitsch HJ, Guldin WO (1983) Heterotopic interhemispheric cortical connections in the rat. Brain Res Bull 10:805–810
Maxwell B, Powell DA, Buchanan SL (1994) Multiple and single unit activity in area 32 (prelimbic region) of the medial prefrontal cortex during Pavlovian heart rate conditioning in the rabbit. Cereb Cortex (in press)
Mesulam M (1978) Tetramethyl benzidine for horseradish peroxidase neurohistochemistry: a non-carcinogenic blue reactionproduct with superior sensitivity for visualizing neural afférents and efferents. J Histochem Cytochem 26:106–117
Mesulam M (1990) Large-scale neurocognitive networks and distributed processing for attention, language, and memory. Ann Neurol 28:597–613
Neafsey EJ (1990) Prefrontal cortical control of the autonomie nervous system: anatomical and physiological observations. Prog Brain Res 85:147–166
Neafsey EJ, Hurley-Guis KM, Arvanitis D (1986) The topographical organization of neurons in the rat medial frontal, insular and olfactory cortex projecting to the solitary nucleus, olfactory bulb, periacqueductal gray and superior colliculus. Brain Res 377:261–270
Pandya DN, Yeterian EH (1990) Prefrontal cortex in relation to other cortical areas in rhesus monkey: architecture and connections. Prog Brain Res: 63–94
Porrino LJ, Goldman-Rakic PS (1982) Brainstem innervation of prefrontal and anterior cingulate cortex in the rhesus monkey revealed by retrograde transport of HRP. J Comp Neurol 205:63–76
Powell DA, Buchanan SL, Gibbs CM (1990) Role of the prefrontal-thalamic axis in classical conditioning. Prog Brain Res 85:433–466
Powell DA, Watson K, Maxwell B (1994) Involvement of subdivisions of the medial prefrontal cortex in learned cardiac adjustments. Behav Neurosci 108:294–307
Reep RL, Goodwin GS, Corwin JV (1990) Topographic organization in the corticocortical connections of medial agranular cortex in rats. J Comp Neurol 294:262–280
Room P, Russchen FT, Groenewegen HJ, Lohman AHM (1985) Efferent connections of the prelimbic (area 32) and the infralimbic (area 25) cortices: an anterograde tracing study in the cat. J Comp Neurol 242:40–55
Rose M (1931) Cytoarchitektonischer Atlas der Grosshirnrinde der Kaninchens. J Psychol Neurol 43:353–440
Rose JE, Woolsey CN (1948) Structure and relations of limbic cortex and anterior thalamic nuclei in rabbit and cat. J Comp Neurol 89:279–348
Rosene DL, Roy NJ, Davis BJ (1986) A cryoprotection method that facilitates cutting frozen sections of whole monkey brains for histological and histochemical processing without freezing artifact. J Histochem Cytochem 34:1301–1315
Saper CB (1982) Convergence of autonomic and limbic connections in the insular cortex of the rat J Comp Neurol 210:163–173
Schwaber JS, Kapp BS, Higgins GA, Rapp PR (1982) Amygdaloid and basal forebrain direct connections with the nucleus of the solitary tract and the dorsal motor nucleus. J Neurosci 2:1424–1438
Sesack SR, Deutch AY, Roth RH, Bunney BS (1989) Topographical organization of the efferent projections of the medial prefrontal cortex in the rat: an anterograde tract-tracing study withPhaseolus vulgaris leucoagglutinin. J. Comp Neurol 290:213–242
Stepniewska I, Kosmal A (1986) Distribution of mediodorsal thalamic afferents originating in the prefrontal association cortex of the dog. Acta Neurobiol Exp 46:311–322
Tehovnik EJ, Spence SJ, Saint-Cyr JA (1989) Efferent projections of the anteromedial cortex of the rat as described byPhaseolus vulgaris leucoagglutinin immunohistochemistry. Behav Brain Res 35:153–162
Uylings HBM, van Eden CG (1990) Qualitative and quantitative comparison of the prefrontal cortex in rat and in primates, including humans. Prog Brain Res 85:31–62
Van Der Kooy D, Koda LY, McGinty JF, Gerfen CR, Bloom FE (1984). The organization of projections from the cortex, amygdala, and hypothalamus to the nucleus of the solitary tract in rat. J Comp Neurol 224:1–24
Vogt BA, Sikes RW, Swadlow HA, Weyland TG (1986) Rabbit cingulate cortex: cytoarchitecture, physiological border with visual cortex, and afferent cortical connections of visual, motor, postsubicular, and intracingulate origin. J Comp Neurol 248:74–94
Wyss JM, Sripanidkulchai K (1984) The topography of the mesencephalic and pontine projections from the cingulate cortex of the rat. Brain Res 293:1–15
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Buchanan, S.L., Thompson, R.H., Maxwell, B.L. et al. Efferent connections of the medial prefrontal cortex in the rabbit. Exp Brain Res 100, 469–483 (1994). https://doi.org/10.1007/BF02738406
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02738406